Comparable to the space race of the 1960s and 70s, a new obsession has gripped the world, globally-the race for the coronavirus vaccine. Countries are investing billions of dollars in pharmaceutical companies and independent research teams alike, to push out a cure, and start mass manufacturing by the end of 2020!
The novel Corona Virus has certainly proved that challenging the elements of nature can have damaging outcomes. The world that currently suffers from the sickness, shares the pain globally. Thousands and millions have already succumbed to the disease, while the rest are trying to win the battle against it. The healthcare industry appears to be in shambles as it tries to provide aid to ailing patients.
However, coping with the disease seems to be quite challenging for healthcare professionals. Not only has the industry got to deploy the services of medical professionals 24*7 but also has to look after the process of Scene Clean at the contaminated zones. Along with this, the industry has to double its efforts to increase the production of masks and other sanitization products. Most importantly, they have to ensure that the vaccines made will have fruitful results.
Countries like the US, European Union, and China have already made deals to receive at least 100 million doses from a few companies as soon as the vaccines hit the market. The US alone invested 50 billion dollars in its ‘Operation Warp Speed’, singling out 6 candidates who are most likely to come out with a vaccine this year. 💉
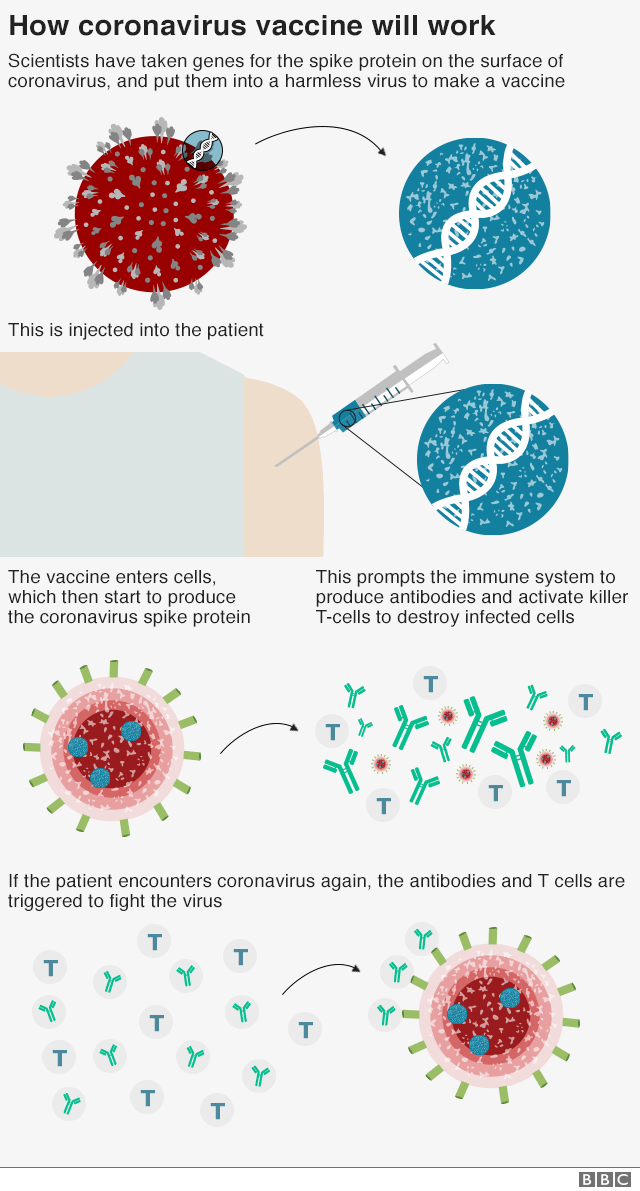
How does the coronavirus vaccine work?

When an antigen (foreign body) enters our system, our body recognizes it as an intruder and immediately launches an attack against it. 🙅 Vaccines work by injecting a substance that mimics the appearance of the real antigen. It’s usually an attenuated (dead or weakened) version of the antigen.
The result is the deployment of an army of defense cells (called lymphocytes) that produce antibodies that fight the antigen and destroy it. 👊 The lymphocytes can read specific proteins on the antigens, which helps them in recognizing whether a molecule is wanted or unwanted.
Apart from the traditional approaches, there are three new types of vaccines being developed for the coronavirus:
1. mRNA-based vaccine
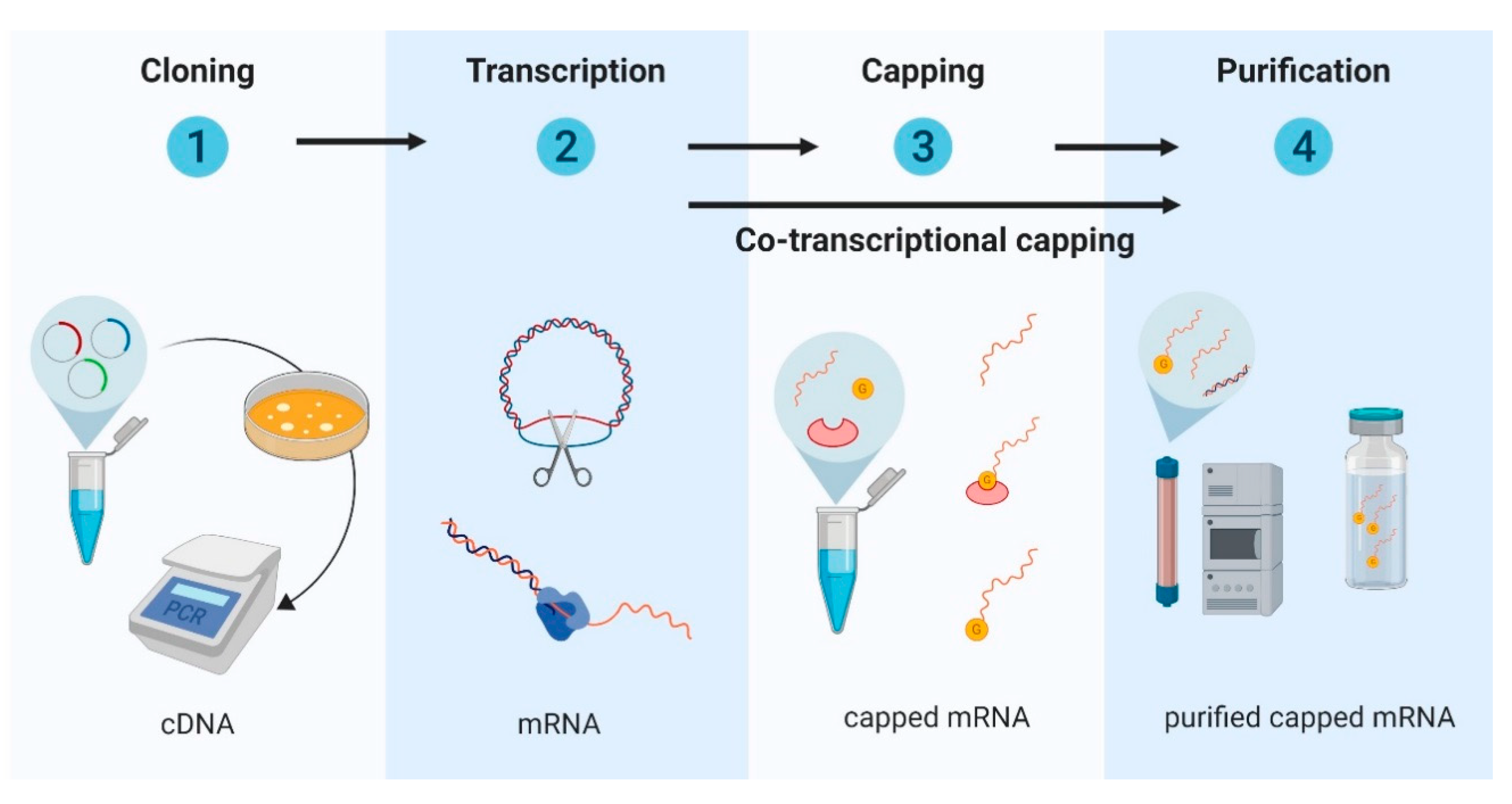
RNA is a material in our cells that contains instructions to make proteins. An mRNA vaccine injects a “code” into cells, instead of an attenuated virus into the body. The “code” then encourages the body to produce a protein that looks like the coronavirus. This look-alike then produces an immune response. The idea is to teach the body what the real invader looks like.
Most companies are currently developing various forms of the mRNA vaccine.
2. DNA-based vaccine

DNA codes for RNA, and RNA codes for proteins. Similar to the mRNA vaccines, DNA-based vaccines also encourage the body to synthesize a molecule that looks like the coronavirus. This generates an immune response.
3. Protein-based vaccine
 Also called as sub-unit vaccines, these vaccines contain a small part of the coronavirus, genetically engineered into another less dangerous virus. Eg. Novavax’s sub-unit vaccine uses a baculovirus. We usually give sub-unit vaccines with an “adjuvant” (a substance that enhances the immune system’s response) to increase the effectiveness of the vaccine.
Also called as sub-unit vaccines, these vaccines contain a small part of the coronavirus, genetically engineered into another less dangerous virus. Eg. Novavax’s sub-unit vaccine uses a baculovirus. We usually give sub-unit vaccines with an “adjuvant” (a substance that enhances the immune system’s response) to increase the effectiveness of the vaccine.
Who are the top contenders in the race for the COVID-19 vaccine?

1. Moderna
Compared to other pharma giants, Moderna is a humble biotech firm headquartered in Massachusetts, USA. They began testing for their mRNA-1273 vaccine as early as January 2020; by February end they had completed their Phase 1 clinical trials.
As of now, they have entered the last leg of the race (Phase 3 clinical trials) and are looking to test the vaccine on over 30,000 adult subjects. If all goes according to plan, Moderna will be well on its way to produce about 500 billion doses by 2021!
2. Pfizer and BioNTech
Pfizer has partnered with German tech company BioNTech to work on a similar mRNA vaccine. The mRNA instructs normal cells to mimic the coronavirus structure, causing the body to produce an immune response.
Pfizer has also entered phase 3 clinical trials, hoping to test a pool of 30,000 subjects.
3. Oxford University and AstraZeneca
Partnering with AstraZeneca is the research team at the University of Oxford. Their vaccine uses a weakened common cold virus (adenovirus) to house some of the coronavirus’ genetic material. This recombinant structure of the vaccine tricks the immune system to fight back.
A single dose of this vaccine provoked an excellent immune response, compared to Moderna’s and Pfizer’s mRNA vaccines that required two doses to show significant results.
4. CanSino
Chinese-based company CanSino is also using adenovirus in its vaccine profile. This technology was used to make the first Ebola Vaccine back in 2017 too.
Currently, the adenovirus-based vaccine has been approved for use by the Chinese military. CanSino will begin their Phase 3 trials soon.
5. Sinopharm
Sinopharm’s vaccine is an inactivated version of the coronavirus. It is being grown in the lab using vereo cells-kidney cells taken from the African green monkey. Sinopharm is testing the potential vaccine in the United Arab Emirates in a Phase 3 trial expected to recruit 15,000 people, as China has too few new cases to be a useful trial site.
6. Novavax
Another type of virus-the baculovirus, known to infect insects-is being used by US pharma company Novavax. Here, spike proteins from the coronavirus are introduced into the baculovirus. This modified baculovirus is then grown in the lab. It produces proteins similar to the coronavirus antigen, which can be injected into the body to produce an immune response. This vaccine is given with an adjuvant called Matrix M, which increases its effectiveness.
The typical time taken to develop a vaccine is usually 10-15 years. But countries have upped their game and research is ongoing at an unprecedented rate. Naturally, one vaccine won’t cut it; the need of the hour is multiple vaccines that will reach all corners of the globe! Cheers to all the frontline workers helping make it happen. 👨”âš•ï¸ğŸ‘©”âš•ï¸
Written and Researched by Shaista Salam